液体波导毛畑管流通池
产品概况
典型的比色皿和流通池光程位于2毫米和10厘米之间,由于机械约束很难开发更长的光程。WPI公司生产的 液体波导毛细管流通池(LWCC)填补了这个空隙。
液体波导毛细管流通池(LWCC)是一种基于光纤技术的流通池,将不同光程(2毫米-500厘米)光学通路 结合少量液体样品(2.4微升-3000微升)检测的检测技术。
LWCC通过光纤连接线可以与基于LED光源的LEDSpec组成流体样品光谱仪,也可通过光纤连接到诸如TIDAS 系统的光谱分析仪或微CCD阵列组成的光谱仪。
在实验室或实际应用的过程控制环境中,LWCC可以在UV/VIS/NIR紫外波长、可见光波长和近红外范围内 完成超低浓度物质的吸光度测量,达到检测样品低浓度的目的。
工作原理
LWCC由熔融石英管外面包被一层低反射指数的多聚物组成,液体样本引导进入毛细管并代表波导核心。 熔融石英毛细管内壁的亲水特征导致信号稳定性高,并很容易清除困陷在流通池中的气泡。
根据BEER定律,物质的吸光度信号与检测的化合物浓度、光程长度和该化合物特定的摩尔吸收系数成正比, 因此,与标准的1厘米光程的比色皿相比,使用WPI公司专利的液体波导技术的50厘米光程的LWCC,测量同样 一个ImAU吸光度信号增强了50倍,而500厘米光程的LWCC则信号灵敏度提高了500倍。
LWCC可直接连接到蠕动泵或用注射器贮满液体进样。
产品特征
1、不同光程长度可选,包括50厘米、100厘米、250厘米和500厘米光程:
2、与1厘米光程比色皿相比,测量结果灵敏50倍到500倍不等;
3、毛细管内径只有0.55毫米,非常适合于少量液体体积的样本测量;
4、内径为2毫米的液体波导毛细管流通池可以用于未经过滤的液体样本测量;
5、标准SMA905连接头用于检测器与流通池的连接:
6、检测光谱范围与LWCC的透光性能相关,而透光性取决于液体样本的固有衰减。使用水的情况下,100厘 米光程LWCC的波长范围从250-720纳米可观察;如果使用500厘米光程LWCC,透光范围就降低到300-700 纳米。然而,将水转换成甲醇作为溶剂时,使用合适的光源和检测器,就可能透过近红外。
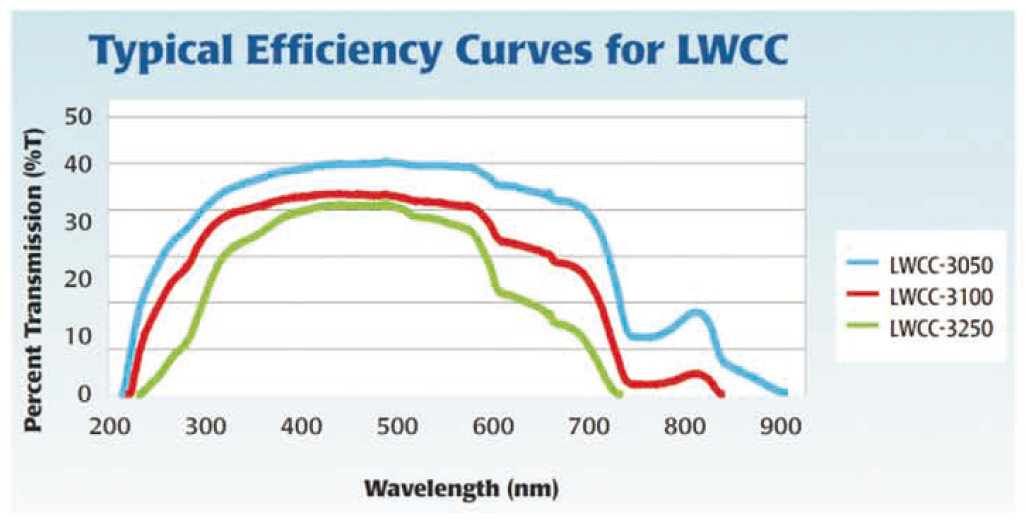

产品分类
1.长光程LWCC


2.微型LWCC


3.多光程LWCC


UltraPath是一款独一无二的高效光谱仪系统,具有2、10、50和200厘米的可选择光程,可在250-730nm或 380-730nm波长范围内进行操作,具有独特的动态量程。
该设备主要应用于对水溶液中超低吸光度物质的检测,它也是一款应用于对精确度和灵敏度要求非常高的 确定分析物光谱带的理想工具,既用在实验室也用在工作场地。
UltraPath是WP1公司与NASA协同开发用于确定海水和淡水中有色溶解有机物光谱带,它也可以用于实验室 和工作场所如大海。CDOM浓度在广阔的海水样本中是低的(在380nm波长时为0.007m-1),而在淡水中是高的 含量(如在380nm波长时为10〜20m-l)。
要解决这些问题,UltraPath系统按照设计要求开发一款坚固的便携设备,能够高灵敏地在一个宽的动态范围 内对不同样本进行测量,并使CDOM在0.002m-1到200m-1浓度范围内250-730nm波长范围内的测量更加可靠。
仪器特征
1、可用于过程控制和海洋学研究;
2、非常坚实的系统,用于实验室和船载测量;
3、方便携带,使用简单;
4、用户可选光程长度:2、10、50和200厘米;
5、灵敏度高和稳定性好;
6、与NASA共同设计用于海水和淡水中有色溶解有机物的测量:
7、从紫外到可见光波段吸光度测量,具有非常灵敏的可延伸动态范围;与标准长光程流通池协同使用并不复杂;
主要应用
一、CDOM-有色溶解有机物测量
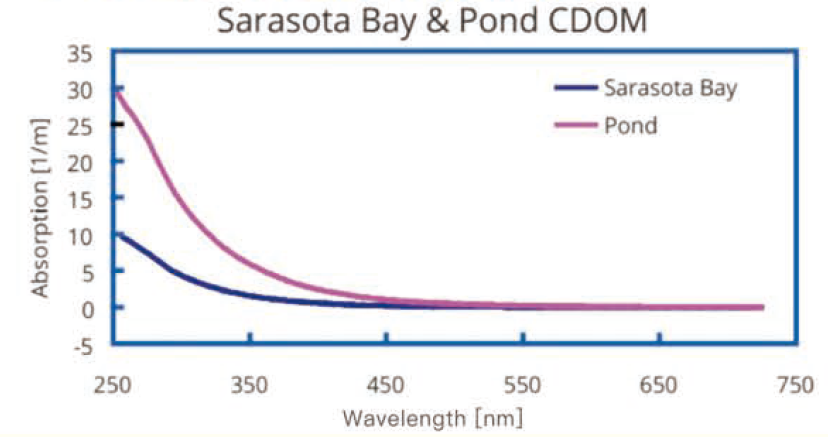
1.使用UltraPath系统测量所得两个典型的吸收光 谱。标记为“Sarasota Bay "是2007年11 月从Sarasota 海湾釆集的-个34PSU盐度的CDOM样本。标记为“Pond"是2007年11月从S arasota的一个地方池塘里收集的高浓度CDOM样本。

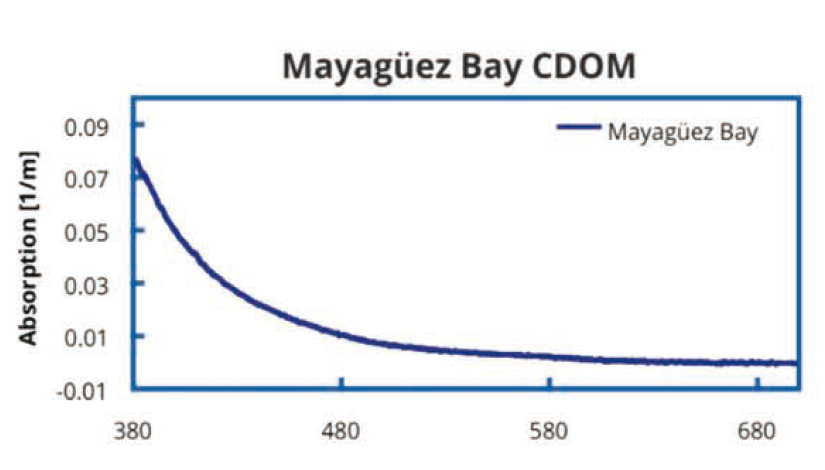
2.标记为 “Mayagiiez Bay”的CDOM样本2001 年 釆集于波多黎各西海岸Mayaguez海湾,该区域海水 具有高盐度、寡营养海水,资料有NASA的Stennis 空间中心友情提供。

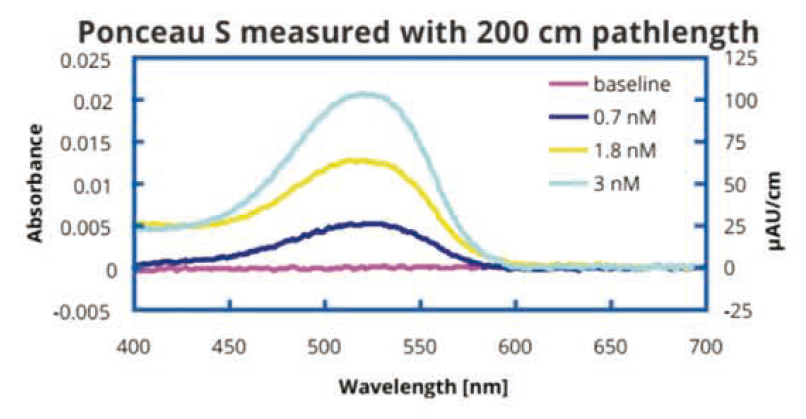
二、QFT-定量滤波技术: 使用U ItraPath的200厘米光程流通池测 量胭脂红S吸光度。胭脂红S溶解在Millipore 的纯水中。

参考文献
LWCC用于海水和天然水研究
[1] Development of a sequential injection analysis device and its application for the determination of Mn(II) in water Taianta 2020, 211:120752 https://doi.Org/10.1016/j.talanta.2020.120752
[2] Concentrations of reactive Mn(III)-L and Mn02 in estuarine and marine waters determined using spectrophotometry and the leuco base, leucoberbelin blue Taianta 2019, 200:91-99
https://doi.Org/10.1016/j.talanta.2019.03.026
[3] Determination of ammonia nitrogen in natural waters: Recent advances and applications Trends in Environmental Analytical Chemistry 2019, 24:e00073 https://doi.Org/10.1016/j.teac.2019.e00073
[4] Mn speciation at nanomolar concentrations with a porphyrin competitive ligand and UV-vis measure¬ments Taianta. 2019, 200:15-21.
https://doi.Org/10.1016/j.talanta.2019.02.069
[5] Attenuation Coefficients of Tubular Conduits for Liquid Phase Absorbance Measurement: Shot Noise Limited Optimum Path Length Anal. Chem. 2019, 91(15):9481-9489 https://doi.org/10.1021/acs.analchem.9b00067
[6] Inherent optical properties and particle characteristics of the sea-surface microlayer Progress in Oceanography 2019, 176:102117
https://doi.Org/10.1016/j.pocean.2019.05.009
[7] Latitudinal Gradient of UV Attenuation Along the Highly Transparent Red Sea Basin Photochem Photobiol. 2019, 95(5):1267-1279.
https://doi.org/10.llll/php.13112
[8] Mobility of electrostatically and sterically stabilized gold nanoparticles (AuNPs) in saturated porous media Environmental Science and Pollution Research 2019, 26(28):29460-29472
https://doi.org/10.1007/sll356-019-06132-8
[9] Hydrography-Driven Variability of Optically Active Constituents of Water in the South Brazilian Bight: Biogeochemical Implications Front. Mar. Sci., Posted 19 November 2019 https://doi.org/10.3389/fmars.2019.00716
[10] The influence of dissolved organic matter on the marine production of carbonyl sulfide (OCS) and carbon disulfide (CS2) in the Peruvian upwelling Ocean Sci., 2019, 15:1071-1090 https://doi.org/10.5194/os-15-1071-2019
[11] Automated syringe-pump-based flow-batch analysis for spectrophotometric determination of trace hexavalent chromium in water samples Microchemical Journal 2019, 145:1135-1142
https://doi.Org/10.1016/j.microc.2018.12.040
[12] Spectrophotometric flow injection determination of dissolved titanium in seawater exploiting in-line nitrilotriacetic acid resin preconcentration and a long path length liquid waveguide capillary cell Analytica Chimica Acta 2019, 1053:54-61
https://doi.Org/10.1016/j.aca.2018.ll.034
[13] Direct emission of nitrous acid (HONO) from gasoline cars in China determined by vehicle chassis dy¬namometer experiments Atmospheric Environment 2017, 169:89-96
https://doi.Org/10.1016/j.atmosenv.2017.07.019
多光程LWCC用于海洋学研究
[1 ] Measurements of CDOM absorption spectra using different Instruments and techniques: A round robin exercise and extensive field data set., Ocean Optics XXII, (2014)
[2] Hydrography of chromophoric dissolved organic matter in the North Atlantic. Deep Sea Research. 2007, Parti,54: 710-731.
[3] Variability of chromophoric organic matter in surface waters of the Atlantic Ocean.
Deep Sea Research. Part II: Topical Studies, 2006, 53(14-16):1666-1684.
[4] Determining CDOM Absorption Spectra in Diverse Coastal Environments Using a Multiple Pathlength, Liquid Core Waveguide System Continental Shelf Research, 2002, 22:9:1301-1310.
[5] System Analyzes Water Samples at Sea, NASA Aerospace Technology Innovation, 2001,9 (5).
[6] Evaluating the influence of CDOM on the remote sensing signal in the Mississippi River Bight. In Eos Transactions AGU Ocean Sciences, 2002. Honolulu, HI, p. 171.
[7] Aparent Optical Properties in Waters Influenced by the Mississippi River, Proceedings of the Seventh Thematic Conference, Remote Sensing for Marine and Coastal Environments, 2002, Miami, FL.
[8] Bio-optical Properties of the Mississippi River Plume and Adjacent Shelf. ASLO Aquatic Sciences, Albuquerque, NM, 2001.
[9] Measuring the absorption of CDOM in the field using a multiple pathlength liquid waveguide system. Ocean Optics XV, pl308, Monaco, October 2000.
LWCC用于大气环境监测研究
[1] Cloud Water Chemistry Associated with Urban Aerosols: Rapid Hydroxyl Radical Formation, Soluble Metals, Fe(II); Fe(III), and Quinones ACS Earth Space Chem. 2020, 4(1):67-76
https://doi.org/10.1021/acsearthspacechem.9b00243
[2] No Evidence for Brown Carbon Formation in Ambient Particles Undergoing Atmospherically Relevant Drying Environ. Sci.: Processes Impacts, 2020, Accepted Manuscript https://doi.org/10.1039/C9EM00457B
[3] PM2.5 Humic-like substances over Xi'an, China: Optical properties, chemical functional group, and source identification Atmospheric Research 2020, 234:104784 https://doi.Org/10.1016/j.atmosres.2019.104784
[4] A semi-automated multi-endpoint reactive oxygen species activity analyzer (SAMERA) for measuring the oxidative potential of ambient PM2.5 aqueous extracts Aerosol Science and Technology 2020, 54(3):304-320
https://doi.org/10.1080/02786826.2019.1693492
[5] Impact of secondary and primary particulate matter (PM) sources on the enhanced light absorption by brown carbon (BrC) particles in central Los Angeles Sci Total Environ. 2020, 705:135902.
https://doi.Org/10.1016/j.scitotenv.2019.135902
[6] Use of iron oxide nanoparticles for immobilizing phosphorus in-situ: Increase in soil reactive surface area and effect on soluble phosphorus Science of the Total Environment 2020, 711:135220
https://doi.Org/10.1016/j.scitotenv.2019.135220
[7] Methylamine* s Effects on Methylglyoxal-Containing Aerosol: Chemical, Physical, and Optical Changes ACS Earth Space Chem. 2019, 3, 9, 1706-1716 https://doi.org/10.1021/acsearthspacechem.9b00103
[8] Automation of radiochemical analysis by flow techniques 一 A review TrAC Trends in Analytical Chemistry 2019, 118:352-367
https://doi.Org/10.1016/j.trac,2019.06.001
[9] Contribution of Charge-Transfer Complexes to Absorptivity of Primary Brown Carbon Aerosol ACS Earth Space Chem. 2019, 3(8):1393-1401 https://doi.org/10.1021/acsearthspacechem.9b00116
[10] Evidence in biomass burning smoke for a light-absorbing aerosol with properties intermediate between brown and black carbon Aerosol Science and Technology 2019, 53(9):976-989
https://doi.org/10.1080/02786826.2019.1617832
[11] Methylamine, s Effects on Methylglyoxal-Containing Aerosol: Chemical, Physical, and Optical Changes ACS Earth Space Chem. 2019, 3(9):1706-1716 https://doi.org/10.1021/acsearthspacechem.9b00103
[12] The secondary nature and light absorption properties of water-soluble organic carbon in the winter of Beijing Earth Environ. Sci. 2019, 330:032035
https://doi.Org/10.1088/1755-1315/330/3/032035
[13] Variations and sources of nitrous acid (HONO) during a severe pollution episode in Beijing in winter 2016 Sci Total Environ. 201% 648:253-262. https://doi.Org/10.1016/j.scitotenv.2018.08.133
[14] Multi-elemental analysis of particulate matter samples collected by a particle-into-liquid sampler Atmospheric Pollution Research 2018, 9(4):747-754 https://doi.Org/10.1016/j.apr.2018.01.006
[15] Absorbing Refractive Index and Direct Radiative Forcing of Atmospheric Brown Carbon over Gangetic Plain ACS Earth Space Chem, 2018, 2(1):31-37 https://doi.org/10.1021/acsearthspacechem.7b00074
[16] Brown Carbon Aerosol in Urban Xi'an, Northwest China: The Composition and Light Absorption Proper¬ties Environ Sci Technol. 2018, 52(12):6825-6833.
https://doi.org/10.1021/acs.est.8b02386
[17] Light absorption properties of brown carbon over the southeastern Tibetan Plateau
Sci Total Environ. 2018, 625:246-251.
https://doi.Org/10.1016/j.scitotenv.2017.12.183
[18] Brown and black carbon in Beijing aerosol: Implications for the effects of brown coating on light ab¬sorption by black carbon Sci Total Environ. 2017, 599-600:1047-1055. https://doi.Org/10.1016/j.scitotenv.2017.05.061
[19] Development and evaluation of a novel monitor for online measurement of iron, manganese, and chro¬mium in ambient particulate matter (PM) Sci Total Environ. 2016, 565:123-131.
https://doi.Org/10.1016/j.scitotenv.2016.04.164














